How To Reduce Compliance Risk for Pharmaceutical and Medical Device Companies
Use Cases and Deployment Scope
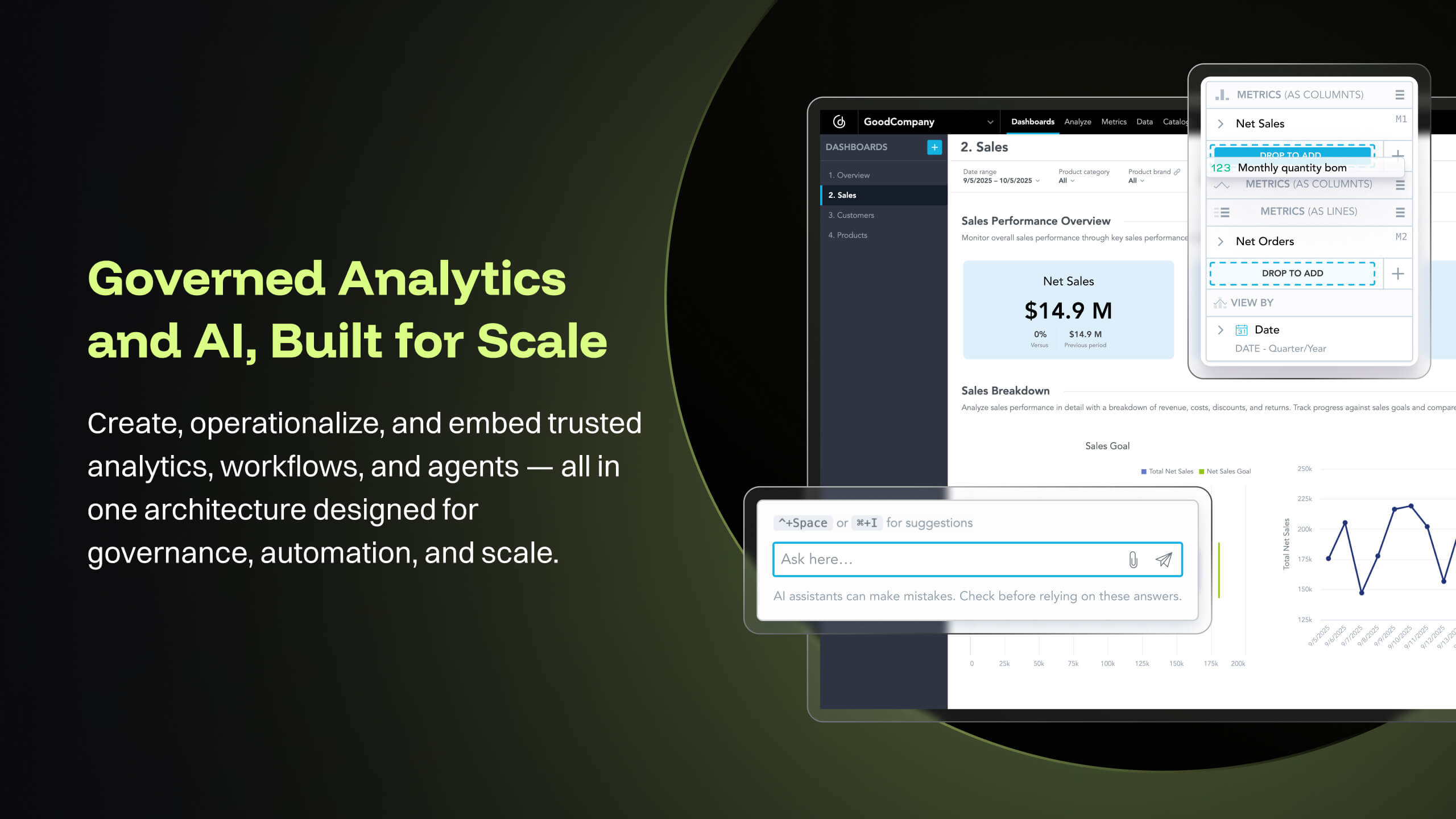
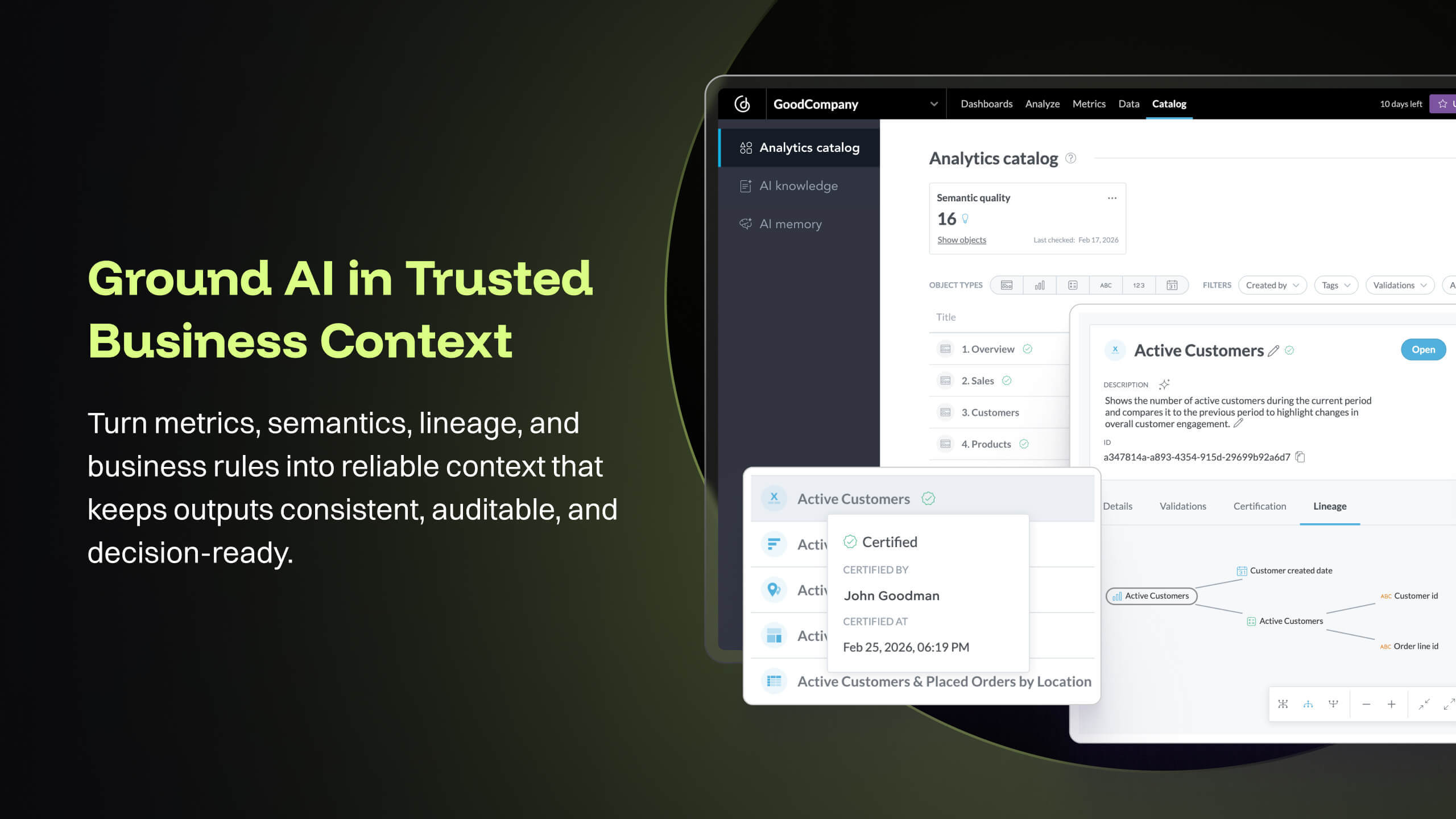
At [...], we are using GoodData Platform to build Embedded Analytics solution which is integrated deep into [...]'s core platform to provide regulatory intelligence, vendor quality intelligence and inspection intelligence for Pharma, pre-market and post-market intelligence in MedTech. This solution involves building key dashboards that provides insights into the world of regulatory affairs that are actionable for our customers. The analytics solution is built on top of a semantic data model that unifies compliance data streams, is well integrated with key objects in the regulatory and quality life-sciences domain, and provides inspection trends and tracks supplier quality risk. Customers at leading Pharma and MedTech companies use these insights and data intelligence to champion quality of their products and ensure safety and compliance.
Pros
- The source datasets are often complex, semi-structured and un-linked to key entities. With GoodData, all of these datasets are unified to serve as a central semantic data model layer, building into a galaxy schema with dimensions, bridge, and facts, which then forms the backbone that powers the [...] data intelligence cloud. Building insights and dashboards become a much easier task once the underlying data model is designed. GoodData enforces certain best practices as a BI tool, which must be adhered to get the true value of the raw data. For e.g. the source FDA dashboard may just show inspection data but the Site Profile dashboard built on GoodData goes beyond the standard information and shows more insight into site risk scores and can be drilled into details. There is blog written on this topic: [...].
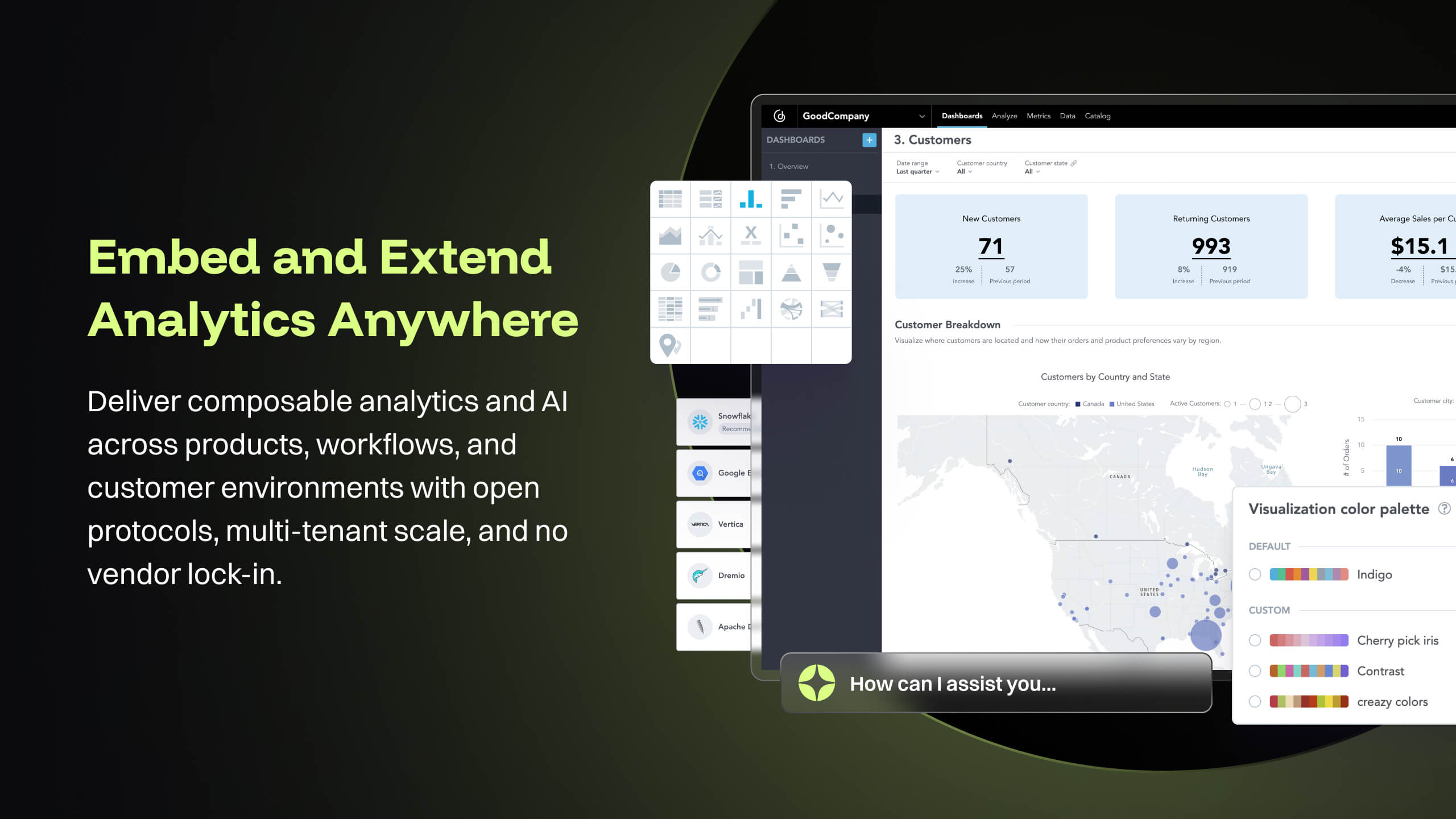
- GoodData provides a rich collection of visualization options that help us create compelling story-telling via dashboards. Being well-prepared for FDA inspections is essential for maintaining product quality, regulatory compliance, and avoiding serious business setbacks. FDA inspections are critical events that can shape a company’s market access and reputation. The FDA itself offers the FDA Data Dashboard, but it doesn’t make every document available. There is a blog written on this topic: [...].
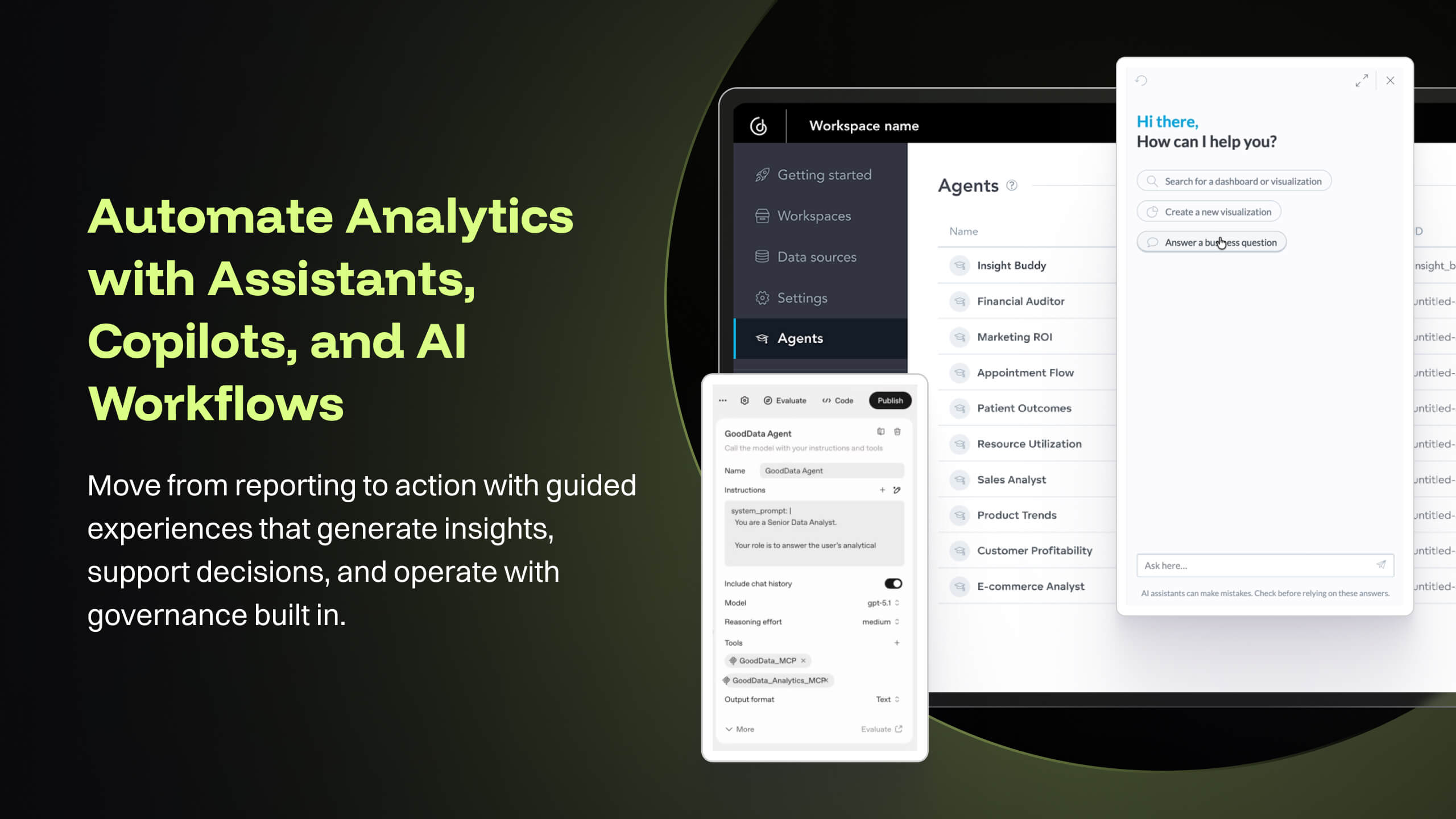
- Medical devices and technologies do not stop evolving after they receive regulatory approval. Once a product hits the market, it faces real-world usage, compliance challenges, and an array of regulatory scrutiny. Managing these postmarket dynamics is critical to a product’s long-term success and patient safety. However, many companies struggle to keep track of relevant events across a product’s markets, from adverse event reports to changing regulations. Postmarket Intelligence developed on GoodData platform enables us to solve that problem. It empowers MedTech companies to efficiently monitor, assess, and act on postmarket data—saving time, improving decision-making, and ensuring compliance with industry standards. Anyone who is used to trying to get the data they need from the various FDA, and other regulatory agency websites, knows that collecting, cleaning, and structuring that data takes hours. And that’s before any analysis can get done. We enable customers to free up time to focus only on the high-value analysis and subsequent recommendations to leadership, rather than wrangling the data.
- The data pipeline refresh that is provided by GoodData Platform is also quite useful from data engineering perspective. The Automated Data Distribution v2 or commonly called as ADD refresh follows a set pattern of identifying the analytical data model through output stage which helps abstract the complex table definition to simpler views that can help with quick rebuild at the data warehouse level while loading the data into GoodData's ADS storage layer. The import first way of loading data into GoodData's cloud storage, followed by querying for any aggregations or metrics on the GoodData analyzer, makes this simple and fast.
- GoodData's latest product i.e. Cloud also offers several good features like Analytics as Code which helps software engineering teams follow a code-first approach to analytics, where building insights, dashboards or even datasets can be done in YAML templates or serviced by REST APIs. This is particularly forward thinking in the modern technology stack and evolving industry requirements. These provide seamless integration options to front-end and backend code, embedded analytics with multiple choices from HTML to React based workloads. At [...], we are currently exploring most of these features while planning for a future migration from Platform to Cloud.
Cons
- GoodData's ADD load for the older product i.e. Platform has some limitations like error logging and debugging is cryptic. If orchestrated via any external tooling like Airflow or Prefect, the REST API based ADD loads sometime fails with HTTP 429 errors indicating as-if there are too many requests but in reality, it is sometimes due to GoodData server busy responding about the outcome of the past execution.
- GoodData Platform's documentation though well-written sometimes lack some of the key information which could make the overall admin process a little too complex to implement. I have encountered issues trying to delete or provision workspaces, where each workspace gets tied to the parent-child hierarchy based segments and that makes the admin process operationally challenging.
- There is a data limitation on the GoodData Platform wherein the maximum allowed characters that can be displayed on a tabular chart for a text field is 10000 characters. At [...], we work with lot of regulatory documents that are often 200+ pages long in their content. We summarize the content of such documents and enrich with our proprietary labels, before finally sharing with customers via dashboards. However, the limitation of characters to default 500 often requires us to ALTER DATATYPE using MAQL to be able to move up to 10000 characters to workaround. At times, even 10000 characters get trimmed and often do not get displayed on mouse-hover. Thus it impacts the customer experience when trying to perceive the dashboard. Note, if the dashboard is downloaded in excel or csv, then and only then the full content is observed.
Return on Investment
- We have launched MedTech Post-market intelligence demonstrating 22+ million post-market medical device events with their analysis of how they are linked to affected products, device problems, patient problems and manufacturers. The ease of being able to process and display such high volume data with intricate joins is done seamlessly within GoodData.
- Another core offering from [...] is MedTech Pre-market intelligence that involves tracking the life-cycle of a product or medical device's licensing journey before it hits the market. This involved processing and supporting 2+ million device records globally and their linkages. Our customers exclaimed "You just did in 15 seconds what took me and my team 3 months to do … and I trust your data more than what we did." For more details, read here:[...]
- [...] is the Intelligence Cloud for quality and regulatory operations, transforming regulatory complexity into trusted, proactive intelligence. We unify global regulatory intelligence, inspection data, and supplier insights so you can predict and prevent compliance risks before they happen. As part of these key offerings, we compute Site Risk scores from supplier analysis standpoint for over 300+ thousand sites and categorize risks in the different department and quality system label issue. These risk score model help customers assess how likely that supplier can get a 483 or warning letter, and what were the top issues reported during their previous inspections. These critical components help customers stay aware of the supplier quality and help them make informed business decisions that saves hundreds of thousands of dollar.
Usability
Alternatives Considered
Tableau Cloud, Looker, Microsoft Power BI, Domo, Highcharts and Metabase
Other Software Used
dbt, Snowflake, Astro by Astronomer