What is Cloudbyz eTMF?
Cloudbyz eTMF solution offers a cloud-based repository of all clinical trial documents including files, images, information, etc. Digitally store, manage, and share all clinical trial-related documents with a centralized overview. Manage essential trial documents, stay inspection ready, and enable real-time visibility for CROs, sponsors, monitors, and other stakeholders in a clinical trial. Cloudbyz eTMF functionality includes-
eTMF Export
Automated & Configurable Metadata
TMF Reference Models
Actionable Notifications
eTMF Locking
Search and Retrieval
eISF Connectivity
Real Time Collaboration
TMF Health Check
Inspection Readiness
- Milestone Tracking
All Cloudbyz solutions are natively built on the Salesforce platform and leverage a common data model to ensure seamless integration between the systems. Additionally, users can leverage the template-based approach to accelerate their trial operations. Their solutions span across the entire life cycle of clinical trials and help customers to stay fully compliant with global industry regulations.
Categories & Use Cases
Screenshots
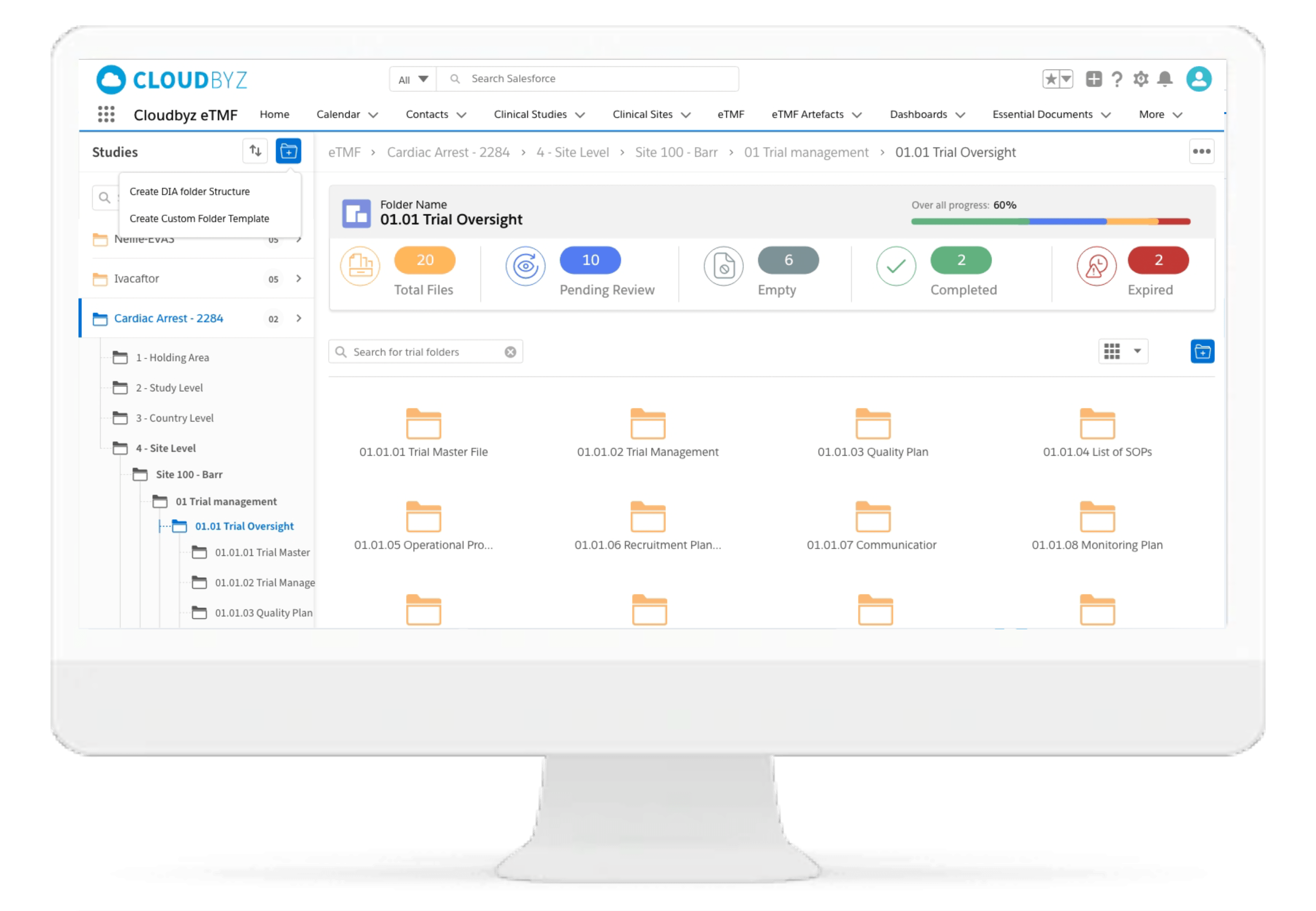
Standard and Custom TMF reference Models
Product Demos
Technical Details
| Deployment Types | SaaS |
|---|---|
| Mobile Application | Apple iOS, Android, Mobile Web |
| Supported Countries | Global |
| Supported Languages | Danish, German, English, Finnish, French, Italian, Japanese, Korean, Dutch, Portuguese, Russian, Spanish, Swedish, Thai, Chinese |




